Aluminum
by Chris Woodford. Last updated: January 21, 2024.
Suppose you had to design the perfect material—what would it be like? You'd probably want it to be plentiful and relatively inexpensive, strong and lightweight, easy to combine with other materials, resistant to heat and corrosion, and a good conductor of electricity. In short, you'd probably come up with a material like aluminum (spelled aluminium in some countries—and that's also the official IUPAC spelling).
It's the commonest metal in Earth's crust (making up ~8 percent of it), the third most plentiful chemical element on our planet (only oxygen and silicon exist in greater quantity), and the second most popular metal for making things (after iron/steel). [4] We all see and use aluminum every day without even thinking about it. Disposable drinks cans are made from it and so is cooking foil. You can find this ghostly gray-white metal in some pretty amazing places, from jet engines in airplanes to the hulls of hi-tech warships. What makes aluminum such a brilliantly useful material? Let's take a closer look!
Photo: Aluminum is a wonderfully weather-proof material. At the Federal Building and U.S. Courthouse, Wheeling, West Virginia, it features prominently in the striking windows and other internal features. Photo by Carol M. Highsmith, courtesy of Photographs in the Carol M. Highsmith Archive, Library of Congress, Prints and Photographs Division.
Sponsored links
Contents
What's aluminum like?
Aluminum is soft, lightweight, fire-proof and heat-resistant, easy to work into new shapes, and able to conduct electricity. It reflects light and heat very effectively and it doesn't rust. It reacts easily with other chemical elements, especially oxygen, and readily forms an outer layer of aluminum oxide if you leave it in the air. We call these things aluminum's physical and chemical properties.
Photo: The experimental aluminum Ford Sable car, produced over 25 years ago in 1995, was 180 kg (400 lbs) lighter than a comparable steel-bodied car and considerably more energy efficient. Today, as fuel economy becomes ever more important, full-bodied aluminum cars are commonplace. A new Ford F-150 truck, with a full aluminum body, is a whopping 39 percent (320kg or 700lbs) lighter than its predecessor, according to the Aluminum Association. But that switch proved to be a mixed bag for Ford. Photo courtesy of US Department of Energy (DOE).

Alloys
Aluminum really comes into its own when you combine it with other metals to make aluminum alloys (an alloy is a metal mixed together with other elements to make a new material with improved properties—it might be stronger or it might melt at a higher temperature).
A few of the metals commonly used to make aluminum alloys include boron, copper, lithium, magnesium, manganese, silicon, tin, and zinc. You mix aluminum with one or more of these depending on the job you're trying to do.
There are several hundred different aluminum alloys. Three of the best known are alnico, made from iron, aluminum, nickel, and cobalt, and used in loudspeaker magnets; duraluminum, mostly made of aluminum and copper, and used in aircraft and automobile parts; and magnox, made of magnesium and aluminum and used in nuclear reactors. [5]

Photo: A typical alnico (aluminum, nickel, cobalt, iron alloy) loudspeaker from an old portable radio. Alnico magnets have now largely been replaced by ceramic (and other kinds of) magnets.
There are so many different aluminum alloys that we need a clear system for organizing and referring to them. First, they're classified as either wrought (hammered or otherwise mechanically shaped) or cast (shaped by being poured into molds). Wrought alloys are resistant to corrosion, conduct heat and electricity very well, are strong but still light, are tough and don't fracture, are very workable, and are easy to recycle. Cast alloys, on the other hand, are easy to cast (flow and fill their molds well), strong, and offer a superior finish. [7]
According to a widely used classification system (the International Alloy Designation System or IADS) introduced by the Aluminum Association, wrought alloys are divided into nine series, numbered with four digits, beginning with a number that defines the main alloying element: [8]
- 1xxx: Almost pure (over 99%) aluminum.
- 2xxx: Copper: High-strength aerospace and automative alloys with less corrosion resistance.
- 3xxx: Manganese: Moderate strength, widely used in roofing materials, home cookware, and heat exchangers, as well as aluminum drinks cans.
- 4xxx: Silicon: Lower melting points, used in automobile engine components, welding and brazing wires.
- 5xxx: Magnesium: High strength and greater corrosion resistance, so widely used in marine applications where resistance to saltwater is crucial, as well as storage tanks and building construction materials.
- 6xxx: Magnesium and silicon: A good compromise between strength and workability, used in housing parts such as aluminum window frames and truck and ship structures.
- 7xxx: Zinc: Extremely strong, used in structural airplane components. The Apple aluminum watch used a 7xxx series alloy.
- 8xxx: Other alloying elements (typically lithium).
- 9xxx: Not assigned.
Cast aluminum alloys are also divided into eight series, but the numbering is slightly different:
- 1xxx: Almost pure (maximum 99%) aluminum.
- 2xxx: Copper.
- 3xxx: Silicon with copper and/or magnesium.
- 4xxx: Silicon.
- 5xxx: Magnesium.
- 6xxx: Not used.
- 7xxx: Zinc.
- 8xxx: Tin.
- 9xxx: Other alloying elements.
Composites
Aluminum can be combined with other materials in a quite different way in composites (hybrid materials made from two or more materials that retain their separate identity without chemically combining, mixing, or dissolving). So, for example, aluminum can act as the "background material" (matrix) in what's called a metal matrix composite (MMC), reinforced with particles of silicon carbide, to make a strong, stiff, lightweight material suitable for a wide variety of aerospace, electronic, and automobile uses—and (crucially) better than aluminum alone.
What's aluminum used for?
Pure aluminum is very soft. If you want to make something stronger but still lightweight, hard-wearing, and able to survive the high temperatures in an airplane or car engine, you mix aluminum and copper. For food packaging, you don't need anything like the same strength, but you do need a material that's easy to shape and seal. You get those qualities by alloying aluminum with magnesium.
Suppose you want to carry electricity over long distances from power plants to homes and factories. You could use copper, which is generally the best conductor (carrier) of electricity, but it's heavy and expensive. Aluminum might be an option, but it doesn't carry electricity so readily. One solution is to make power cables from aluminum alloyed with boron, which conducts electricity almost as well as copper but is a great deal lighter and less droopy on hot days. (Typically, aluminum-boron alloys contain 90–97 percent aluminum and 3–10 percent boron.) [6]

Chart: Aluminum consumption in the United States. Transportation (planes, ships, trucks, and cars) is now by far the biggest single use for the metal and its alloys. Source: US Geological Survey, Mineral Commodity Summaries: Aluminum. January 2023.
How is aluminum made?
Aluminum reacts so readily with oxygen that you never naturally find it in its pure form. Instead, compounds of aluminum exist in huge quantities in Earth's crust as an ore (raw rocky material) called bauxite. This is the common name for hydrated alumina, a substance typically made from about two thirds aluminum oxide (chemical formula Al2O3) with one third water molecules (H2O) locked into its crystal structure.

Photo: A hill of bauxite awaiting processing in Sheffield, Alabama c.1942. Photo by Jack Delano, Office of War Information, courtesy of Library of Congress, Prints and Photographs Division.
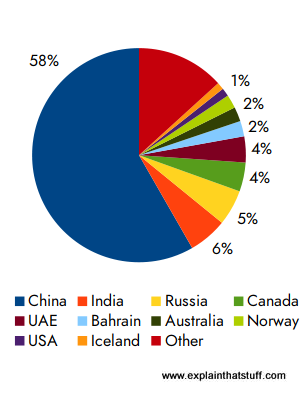
Depending on where on Earth it's found, bauxite also contains a range of different impurities such as iron oxide, silicon oxide, and titanium oxide. The world currently has about 55–75 billion tons of bauxite resources—enough to meet demand "well into the future" (according to the US Geological Survey's Mineral Commodity Summaries, January 2023).

Photo: Ready for recycling: These squashed mats of aluminum cans are called biscuits. They're ready to melt down and recycle. According to the Aluminum Association, nearly 70 percent of the aluminum ever mined is still in use today, thanks to effective recycling programs. It's much cheaper and more environmentally friendly to recycle used aluminum than to dig bauxite from the ground and process it: recycling saves about 95 percent of the energy that would be needed to make brand new aluminum. Photo courtesy of US Air Force.
If you want to turn bauxite into aluminum to make useful things like cans, cooking foil, and space rockets, you've got to get rid of the impurities and the water and split the aluminum atoms from the oxygen atoms they're locked onto. So making aluminum is actually a multi-stage process.
First, you dig the bauxite from the ground, crush it up, dry it (if it contains too much water), and purify it to leave just the aluminum oxide. Then you use an electrical technique called electrolysis to split this into aluminum and oxygen. (Electrolysis is the opposite to what happens inside a battery. In a battery, you have two different metal connections inserted into a chemical compound and complete a circuit between them to generate electricity. In electrolysis, you pass electricity, via two metal connections, into a chemical compound, which then gradually splits apart into its atoms.) Once separated out, the pure aluminum is cast into blocks known as ingots, which can be worked or shaped or used as a raw material for making aluminum alloys.
Making usable, shiny aluminum from rocky lumps of bauxite that you've dug from the ground is a lengthy, dirty, incredibly energy-intensive process. That's why the aluminum industry is so keen on recycling things like used drink cans. It's far quicker, cheaper, and easier to melt these down and reuse them than it is to process bauxite. It's also much better for the environment because it saves a huge amount of energy.

Chart: Why recycling aluminum makes sense. The amount of energy it takes to recycle metal for reuse (orange bars) is a fraction of what it takes to produce virgin metal in the first place (blue bars), but the difference is much greater for aluminum (center) than for either steel (left) or copper (right) because it's so hard to extract and refine aluminum in the first place. Data source: "Table 7.11 Embodied energy of selected materials" in Energy and Carbon Emissions by Nicola Terry, UIT Cambridge, 2011, based on data from the Inventory of Carbon and Energy (ICE) by the Sustainable Energy Research Team, University of Bath.
Sponsored links
A brief history of aluminum

Photo: Building an aluminum boat. This high-speed aluminum boat, known as the Littoral Surface Craft-Experimental (LSC-X) or X-Craft, is shown here during construction in Freeland, Washington. Photo by John F. Williams courtesy of US Navy and Wikimedia Commons.
Who discovered aluminum, how, and when? Here's the story as it happened...
- 1746: German chemist Andreas Marggraf (1709–1782) realizes that alum (a natural aluminum compound used for dying textiles since ancient times) contains an unknown metal. It's aluminum, of course, but he doesn't know that.
- 1809: English chemist Sir Humphry Davy (1778–1829) names this metal "alumium" and (later) "aluminium", but is unable to separate it out.
- 1825: Danish chemist and electrical pioneer Hans Christian Øersted (1777–1851) turns aluminum oxide into aluminum chloride and then uses potassium to turn the chloride into pure aluminum. Unfortunately, he cannot repeat the trick a second time!
- 1827: German chemist Friedrich Wöhler (1800–1882) also makes a small quantity of aluminum by heating aluminum oxide with potassium metal.
- 1855: French chemist Henri Sainte-Claire Deville (1818–1881) uses sodium to separate out aluminum. Since sodium is cheaper and easier to obtain than potassium, Deville is able to produce more aluminum—enough to make an ingot. He puts this on display at a public exhibition in Paris, France. Deville's new method means aluminum starts to become more widely available and the price begins to fall.
- 1886: Working independently, the American team of Charles Martin Hall (1863–1914) and his sister Julia Brainerd Hall (1859–1925) and Frenchman Paul-Louis-Toussaint Héroult (1863–1914) discover the modern method of splitting aluminum oxide with electrolysis to make pure aluminum. Their highly efficient technique, known as the Hall-Héroult process, is still used to produce most of the world's aluminum today.
- 1888: Austrian chemist Karl Bayer (1847–1904) finds a less expensive way of turning bauxite into aluminum oxide—the raw material needed for the Hall-Héroult process. Together, the Bayer and Hall-Héroult processes drastically reduce the price of aluminum, enabling the metal to be used in much greater quantities.
- 1893: Studebaker launches an aluminum farm wagon for the Chicago World's Columbian Exposition.
- 1899: A Dürkopp sports car with an aluminum body is unveiled at the Berlin International Motor Show. A few years later, the Pierce Arrow Motor Car Company produces its cars with cast aluminum bodies.
- 1901: Motor pioneer Carl Benz produces the first aluminum car engine.
- Early 1900s: First aluminum recycling programs.
- 1913: Aluminum foil first produced.
- 1920s: Modern aluminum alloys begin to appear.
- 1925: American Chemical Society officially changes the name from "aluminium" to "aluminum" in the United States.
- 1946: Aluminum is used for the bodywork of the lightweight, mass-produced Panhard Dyna X.
- 1957: The first aluminum power lines are introduced.
- 1959: Coors produces the first all-aluminum drinks can.
- 1975: Daniel Cudzik invents the stay-on ring-pull tab for drinks cans.
- 1990: The International Union of Pure and Applied Chemistry (IUPAC) officially adopts "aluminium" as its spelling.
- 1994: The Audi A8 sets new standards in lightweight car production with an aluminum body framework weighing just 249kg (almost half the weight of a comparable steel shell).
- 2015: Ford launches an all-aluminum bodied version of its hugely popular F-150 truck.