
Concrete and reinforced concrete
by Chris Woodford. Last updated: June 9, 2022.
Stonehenge in England, the Great Pyramid of Giza, the Peruvian citadel at Machu Picchu—three wondrous examples of how stone structures can last hundreds or even thousands of years. But though stone is one of the oldest and most durable building materials, it isn't exactly easy to work with. It's heavy, hard to transport, and usually comes in giant chunks, which have to be laboriously cut to shape. Wouldn't it be great if there were a recipe for stone—a kind of gooey cake mixture we could throw together wherever it was needed, simply pressing it into molds to make buildings and structures of any shape or size?
Well that kind of "liquid stone" really does exist: we call it concrete. Although it sometimes gets a bad press, because many people associate it with brutal urban architecture from the mid-20th century, concrete is the great, unsung hero of the modern, material world. From the Hoover Dam to the Sydney Opera House, you'll find it in the world's tallest skyscrapers, biggest bridges, longest highways, deepest tunnels, and quite probably even under the floor in your own humble little home. Concrete is pretty amazing stuff, but what is it and how exactly does it work? Let's take a closer look!
Photo: Concrete is the power behind virtually every modern building and major structure—but it's not as ugly as many people suppose. This is the 12-arch Calstock Viaduct that carries a railroad over the River Tamar in Cornwall, England. Although it looks as elegant as old stone, it is in fact made from concrete blocks that were precast on site, and was completed in 1908.
Sponsored links
Contents
What is concrete?
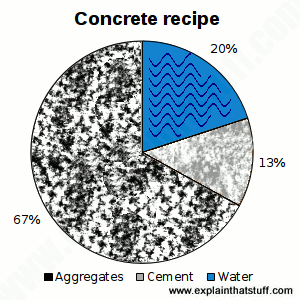
Chart: Concrete recipe: The ingredients of a typical mix.
The word "concrete" comes from a Latin word, concretus, meaning to grow together—and that's exactly what it does when you combine its three ingredients, which are:
- A mixture of coarse and fine aggregates (sand, gravel, stones, larger bits of crushed rock, recycled glass, bits of old recycled concrete, and pretty much anything equivalent)—typically 60–75 percent.
- Cement (the everyday name for calcium silicates and aluminates)—typically 10–15 percent.
- Water—typically 15–20 percent.
Thrown together and mixed well, these simple ingredients make a composite, which is the name we give to a hybrid material that's better in some important way than the materials from which it's made. In the case of concrete, the "important" thing is that it's strong, hard, and durable. Thinking of concrete as a composite material, the cement hydrate is the background, binding material (technically called the "matrix") to which the sand and gravel add extra strength (the "reinforcement").

Photo: Concrete composite: Look closely at this concrete and you can clearly see how it's working: the lighter colored aggregate (stones of various shapes and sizes, which acts as the reinforcement) is bound together by the darker colored cement (the matrix). Not all concrete looks as rough as this, however; I had to look around quite hard to find this example in a concrete post near my home.
How does concrete form from ingredients that are nothing like the final product? When you add water to cement, crystals of cement hydrate (technically, calcium-silica-hydrate) start to grow, which lock the sand and gravel tightly together. It's this gradual crystal formation that gives concrete its strength, rather than the simple fact that it's drying out. Indeed, the reason you have to keep wetting concrete for several days, as it sets, is to "power" the chemical reactions that hydrate the cement. The mushy slushy mixture that tumbles from your concrete mixer gradually turns much harder than the materials from which it's formed. "Liquid stone" becomes stone for real—well, artificial stone, at least. And by "gradually," I really do mean gradually: concrete hardens in hours, gets properly hard after about a month, but continues to harden and strengthen for at least five years after that.
An interesting fact, from recent scientific studies of concrete, is that the "crystals" inside it aren't really crystals at all: they're not well-ordered and perfectly regular, as crystals are supposed to be, but actually have some of the random structure you find in materials like glass (scientifically known as amorphous solids). Concrete contains quite a bit of trapped air (as much as 5–10 percent), because there is some space around the open, three-dimensional structure of cement hydrate crystals and the sand and gravel trapped between them. And that, in turn, explains why concrete can bend and flex, stretch and compress (just a little bit, anyway).
Just like any recipe, you can vary the mixture for concrete somewhat (more water, perhaps, more aggregates, or even chemicals of different kinds) to produce concrete that flows faster, sets harder or more quickly, weathers better, or has a particular color or appearance. Adding a pigment called titanium dioxide, for example, is a simple way of making concrete bright and white—a million miles from the drab gray stuff that gives concrete car parks a bad name. Another variant is aerated concrete, which looks a bit like a very hard sponge with masses of tiny air pockets inside. These allow the concrete to expand and contract in hot and cold weather without fatally cracking and also make it an excellent heat insulating material.

Photo: When concrete is sprayed from a hose at high-speed, instead of slowly laid from a concrete mixer, it's called shotcrete. Left: How shocrete is applied onto a framework of rebar. Photo courtesy of US NAVFAC and Wikimedia Commons. Right: An example of how shotcrete is used to reinforce the entrance to an old mine. Photo courtesy of US EPA and Wikimedia Commons.
Why is concrete such a popular building material?
In cities, at least, concrete is everywhere you look—and it's not hard to understand why. It's easy to make from cheap and readily available ingredients, easy to pour into molds and make into all kinds of shapes (because it starts life a very viscous liquid), and it's both fireproof and (relatively) waterproof. But the main reason it's so widely used in buildings is that it's extremely strong in compression: you can squeeze it or stand a great deal of weight on it. It's widely used in walls and foundations (the vertical supports, in other words) because it's great for resisting weight piled on top. Unfortunately, concrete's very big drawback is that it's about 10 times weaker in tension than in compression. It cracks or snaps easily if you bend or stretch it, unless you reinforce it with steel inside, so it's not much use in horizontal beams. Although concrete looks heavy and monolithic, it's actually much lighter than you might suppose: it's about a fifth as dense as lead, a third as dense as steel, 10 percent less dense than aluminum, and only fractionally more dense than glass.
Although concrete is often mixed on-site and formed into whatever shapes are needed at the time, it can also be supplied in precast "modules"; blocks, beams, wall sections, pavements, and cladding can all be made this way. Giant, modern segmental bridges, for example, are often quickly and inexpensively assembled from identical concrete sections that have been precast in a factory and shipped to their final location. That makes them quicker and easier to construct than if the entire bridge had to be cast in-situ, which is much harder to do in the middle of a river, for example, or in adverse weather conditions. Another option is to make concrete structures that combine some precast sections with other sections formed on-site.

Artwork: Concrete ideas: Thomas Edison immediately understood the brilliance of concrete as a material for making "instant" buildings. In the early years of the 20th century, he devised this method for making "single-pour" concrete houses that could be mass-produced inexpensively in very large quantities. Concrete, from a pair of mixers (blue), is fed to a tank (red), agitated (green), and then carried by an auger screw (orange) to the top of a huge, three-dimensional mold. Poured through the mold, it forms the walls, floors, and roof of the building—and even some of the fittings (like bathtubs) inside! Unfortunately, the idea never caught on. Artwork from US Patent 1,219,272: Process of constructing concrete buildings by Thomas Edison, March 13, 1917, courtesy of US Patent and Trademark Office.
Reinforced concrete
As we've already seen, concrete is a composite material—a cement matrix with aggregates for reinforcement—that works well in compression, but not in tension. We can solve that problem by casting wet concrete around strong, steel reinforcing bars (tied together to make a cage). When the concrete sets and hardens around the bars, we get a new composite material, reinforced concrete (also called reinforced cement concrete or RCC), that works well in either tension or compression: the concrete resists squeezing (provides the compressive strength), while the steel resists bending and stretching (provides the tensile strength). In effect, reinforced concrete is using one composite material inside another: concrete becomes the matrix while steel bars or wires provide the reinforcement.
The steel bars (known as rebar, short for reinforcing bar) are typically made from twisted strands with nobbles or ridges on them that anchor them firmly inside the concrete without any risk of slipping around inside it. Theoretically, we could use all kinds of materials to reinforce concrete. Generally, we use steel because it expands and contracts in the heat and cold roughly as much as concrete itself, which means it won't crack the concrete that surrounds it as another material might if it expanded more or less. Sometimes other materials are used, however, including various kinds of plastics.

Photo: "Liquid stone" to go—pouring concrete from a mixing truck. These construction workers from the US Navy are spreading wet concrete from a truck onto rebar (a grid of steel reinforcing bars). When the concrete sets, the steel bars will give it added strength: concrete plus steel equals reinforced concrete. Picture by Lt. Edward Miller, courtesy of US Navy and Wikimedia Commons.
Prestressed concrete
Although reinforced concrete is generally a better construction material than the ordinary stuff, it's still brittle and liable to crack: in tension, reinforced concrete can fail in spite of its steel reinforcement, letting water in, which then causes the concrete to fail and the rebar to rust. The solution is to put reinforced concrete permanently into compression by prestressing it (also called pretensioning). So instead of putting steel bars into wet concrete as they are, we tension (pull on) them first. As the concrete sets, the taut bars pull inward, compressing the concrete and making it stronger. Alternatively, rebars in reinforced concrete can be stressed after it starts to harden, which is known as poststressing (posttensioning). Either way, keeping concrete in compression is a cunning trick that helps to stop it cracking (and stops cracks from spreading if they do form). Another advantage is that it's possible to use less prestressed or poststressed concrete or smaller, more slender pieces to carry the same loads, compared to ordinary, reinforced concrete.

Photo: Science runs through concrete—how it sets, why it's strong, and why we use it. This concrete word is one of the details on the Onondaga County War Memorial in Syracuse, New York. Credit: Photographs in the Carol M. Highsmith Archive, Library of Congress, Prints and Photographs Division.




