
Fire extinguishers
by Chris Woodford. Last updated: May 25, 2024.
Fire is one of humankind's oldest discoveries; it's also one of our biggest threats. A fire can destroy in a matter of minutes a home or business that has taken decades to establish. That's why methods of putting out fires are so important. Many buildings are equipped with fire extinguishers, but why are there so many different kinds? What do they do to a fire? And how exactly do they work?
Photo: The characteristic blast of "frozen snow" from a carbon dioxide (CO2) extinguisher. Note how the firefighter wears protective gloves and avoids touching the black end of the extinguisher horn, which gets very cold. Photo by Christopher O'Grady courtesy of US Navy.
Sponsored links
Contents
Fire is dangerous!
Before we start, here's something to note: fire is extremely dangerous. Never ever play with fires or anything that can trigger a fire. You could put your life in danger and risk the lives of other people.
Never play with fire-fighting equipment like fire extinguishers and hoses. They're designed to save lives in emergencies, not to set off like toys. If you set off fire alarms as a prank, people are more likely to ignore them when they go off for real, and someone might die as a result. What if that person was a friend of yours or someone in your family?
If you want to try out a fire extinguisher, go visit your local fire department or arrange for them to come to your school. They'll often be happy to oblige. Many fire departments have open days when you can look around, peek inside a fire engine, and even slide down the fire station poles! Keep an eye out in the local media for events like this.

Photo: Fire fighters, like this one from the US Navy, are always happy to show you how to use fire extinguishers the correct way. Photo by Rachel McMarr courtesy of US Navy and Wikimedia Commons.
What is fire?

Photo: Putting out a fire caused, by a mortar attack, with a carbon dioxide (CO2) extinguisher. The white cloud coming from the horn is formed when liquid carbon dioxide stored under pressure in the extinguisher turns back to a freezing cold gas. Photo by Sgt. Jeremiah Johnson courtesy of US Army.
Ask most people what a fire is and they'll tell you it's something frightening and destructive involving flames. But to a scientist, a fire is something much more precise. A fire is actually a chemical reaction called combustion. When combustion happens, substances like wood, paper, oil, or coal (all of which are made from chemicals, even if you don't immediately think of them that way) combine with oxygen in the air to produce water, carbon dioxide, waste gases that make air pollution—and an awful lot of heat. Combustion doesn't normally happen all by itself: things don't burst into flames without help. It usually takes some activation energy (provided by a spark or a match, the heat of the sun, or an overheating machine) to kick off the reaction. Once combustion is underway, the fire seems to continue all by itself.
Breaking the fire triangle
That's not quite true. Fire happens when three things are in the same place at the same time:
- Fuel (something to burn—such as wood or coal).
- Oxygen (usually from the air).
- Heat.
A fire can burn when all these things are present; it will stop when at least one of them is removed. As any fire-fighter will tell you, putting out a fire involves breaking the fire triangle—which means removing either the fuel, the heat, or the oxygen. Suppose a fire breaks out in a pan on top of your cooker, the first thing you normally do is switch off the heat. If that doesn't work, you might soak a towel with water and place it very carefully over the pan (or, better still, use a fire blanket). The towel is designed to block off the supply of oxygen to the fire (the water stops the towel from catching fire and making things worse). Every fire-fighting technique you can think of involves removing heat, oxygen, or fuel—sometimes more than one of those things at the same time. Fire extinguishers work by removing heat, air, or both.
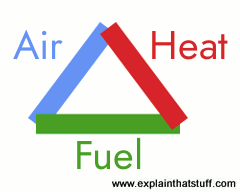
Artwork: You need to to take away one or more of heat, air (oxygen), or fuel to break the triangle and put the fire.
Although professional firefighters have other, more comprehensive ways of understanding fires (including a more complex idea called the fire tetrahedron), the fire triangle is still probably the simplest and most useful rule of thumb for the rest of us.
Types of fire extinguishers
There are four main types of extinguishers and they work in slightly different ways:
- Water extinguishers, which are the most common, are essentially tanks full of water, often with nitrogen or carbon dioxide as the propellant to make them come out. Water extinguishers work mainly by removing heat from the fire, though they also help to cut off a fire's oxygen supply.
- Dry powder extinguishers are tanks of dry powder with compressed nitrogen as the propellant. In extinguishers like this, it's the composition of the chemical (rather than the mechanical design of the extinguisher) that really counts The powder is a specially designed mixture that absorbs heat, melts, and coats the fuel, stopping it from making flammable vapors and blocking out oxygen, so it's helping to tackle two sides of the fire triangle at once. The most widely used powder in extinguishers is monoammonium phosphate; other powder ingredients include the metal alkali salts sodium bicarbonate (baking soda) and potassium bicarbonate (similar to sodium bicarbonate), though these are less effective on things like wood and paper fires.
- Foam extinguishers are tanks of water and foam with compressed nitrogen as the propellant. They work by smothering the fire: when you spread a thin layer of foam over a fire, you cut the fuel off from the oxygen around it. Foam extinguishers also help to absorb heat, since the cool foam they release contains a lot of water.
- Carbon dioxide (CO2) extinguishers contain a mixture of liquid and gaseous carbon dioxide (a nonflammable gas). CO2 is normally a gas at room temperature and pressure. It has to be stored under high pressure to make it a liquid. When you release the pressure, the gas expands enormously and makes a huge white jet. CO2 attacks the fire triangle in two ways: it smothers the oxygen and, when it turns from a liquid back to a gas, it "sucks" in a massive amount of heat from its surroundings (the latent heat of vaporization), which cools whatever you spray it on by removing heat.

Artwork: The four main types of extinguishers and how they break the fire triangle. 1) Water extinguishers work mainly by cooling (removing heat), but a dense spray of water droplets also helps to cut off oxygen. 2) Dry powder extinguishers soak up heat, melt on the fuel, and cut off oxygen, but they also neutralize the fuel (cutting it off from the fire). 3) Foam extinguishers typically float a thin layer of foam over something like a burning liquid, so cutting off oxygen. They also cool a fire. 4) CO2 extinguishers mostly work by cooling a fire, but they also help to cut off its oxygen.
That classifies extinguishers by what they contain. You'll also find fire extinguishers classified by the types of fires you can use them on. This gives us five different kinds:
- A: Green: For wood, cloth, and paper.
- B: Red: For combustible and flammable liquids such as oil, gasoline, and paint.
- C: Blue: For electrical equipment and tools.
- D: Orange:: For flammable metals.
- K: Black: For animal or vegetable oils or cooking fats.
It's important always to use the right extinguisher for the fire. Using the wrong extinguisher can put your life in danger and make the fire worse. For example, you must never use water extinguishers on electrical fires because you could electrocute yourself and the people nearby. If you're in the slightest doubt about tackling a fire, leave it alone and get yourself to safety. Once you've done that, call the fire department.








