Stirling engines
by Chris Woodford. Last updated: March 26, 2023.
Engines have been powering our world since the Industrial Revolution: first, dirty coal-powered steam engines, then cleaner and more efficient gasoline engines, and more recently jet engines in airplanes. The basic concept of an engine—something that exploits the difference between a high temperature and a low one—hasn't changed in a couple of hundred years, although occasionally people do still come up with minor improvements that make the process slightly faster or more efficient. One engine you may have heard a lot about recently is the Stirling Engine, which is a little bit like a steam engine that uses no steam! Instead, it heats, cools, and recycles the same air or gas over and over again to produce useful power that can drive a machine. Teamed up with solar power and other new technologies, Stirling engines sound like cutting-edge technology, but they've actually been around since 1816. Let's take a closer look at how they work!
Photo: Small, compact displacement Stirling engines like this one can run from tiny heat differences—even by resting on someone's hands and running off the heat they contain. Photo courtesy of NASA Glenn Research Center and Internet Archive.
Sponsored links
Contents
What is an engine?
Engines that drive vehicles or factory machines are examples of what scientists call heat engines. They burn an energy-rich fuel (coal, gasoline, or something else) to release heat energy, which is used to make a gas expand and cool, push a piston, turn a wheel, and drive the machine.
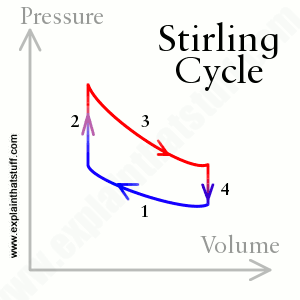
Engines come in two basic types: external combustion engines (for example, steam engines) burn the fuel in one place and produce the power in another part of the same machine; internal combustion engines (such as car engines) burn the fuel and make the power in exactly the same place (in a car, it all happens in the super-strong metal cylinders). Both types of engines rely on heat energy making a gas expand and then cool down. The greater the temperature difference (between the gas at its hottest and its coldest), the better the engine works. The theory of how an engine works is based on the science of thermodynamics (literally, "how heat moves") and on a theoretical model of how ideal engines expand, contract, heat, and cool a gas in a series of steps called a cycle.
Good and bad engines
Before we can discover what's so good about Stirling engines, it helps if we know what's so bad about steam engines. How do they work? You have a coal fire that heats water until it boils and makes steam. The steam travels down a pipe to a cylinder, through an open, entry valve, where it pushes a piston and drives a wheel. The entry valve then closes and an exit valve opens. The momentum of the wheel makes the piston return into the cylinder, where it pushes the cooled, unwanted steam out through the exit and away up the smokestack (chimney).

Photo: Steam engines, such as the one in this locomotive, are examples of external combustion engines. The fire that provides the energy by combustion (1) is outside (external to) the cylinder where heat energy is turned into mechanical power (3). In between the two, there's a boiler (2) that turns the heat energy into steam. The steam acts as a heat-transporting fluid, pushing a piston (4) that moves the wheels with a crank (5) and propels the train (6). Steam and heat energy are constantly being ejected from the smokestack (7), making this a particularly inefficient and inconvenient way to power a moving machine. But it was fine in the days when coal was plentiful and no-one much cared about damaging the planet.
There are many problems with steam engines, but here are four of the more obvious. First, the boiler that makes the steam operates at high pressure and there is a risk that it could explode (boiler explosions were a major problem with the very early steam engines). Second, the boiler is generally some distance from the cylinder, so energy is lost getting the heat from one to the other. Third, the steam leaving the smokestack is still quite hot, so it contains wasted energy. Fourth, because the steam is ejected from the cylinder each time the piston pushes, the engine has to consume huge amounts of water as well as fuel. (That's why steam locomotives have to keep stopping at track-side water tanks.)
What is a Stirling engine?
Can we design an engine that overcomes these problems? Suppose we get rid of the boiler (which would solve the risk of explosions) and use the heat from the fire to power the engine directly. Then, instead of using steam to move heat energy from the fire to the cylinder, why not put the cylinder nearer to the fire and use ordinary air (or some other simple gas) to move the heat energy between them? (This is why Stirling engines are sometimes called hot-air engines.) If we seal this air in a closed pipe, so the same air moves back and forth over and over again, picking up energy from the fire and releasing it in the cylinder, we solve the problem of the engine needing a constant supply of water. Finally, why not add some sort of heat exchanger so that as the hot air passes back and forth, it's energy is retained inside the machine and recycled to improve the overall efficiency. These are the essential ways in which a Stirling engine improves on a steam engine. You'll sometimes see Stirling engines described as "closed-cycle, regenerative heat engines," which is a very terse way of saying what we just said: closed-cycle means they use a sealed volume of gas to move heat back and forth, over and over again, through a series of endlessly repeated steps; regenerative simply means that they use heat exchangers to retain some of the heat that would otherwise be lost on each cycle (blown uselessly up the smokestack, as it is in a steam engine).
Simple or complex?
Some people say Stirling engines are simple. If that's true, it's true in the same way that the great equations of physics (like E = mc2) are simple: they're simple on the surface, but richer, more complex, and potentially very confusing indeed until you really figure them out. I think it's safer to think of Stirling engines as complex: lots of very poor videos on YouTube show how easy it is to "explain" them in a very incomplete and unsatisfactory way. To my mind, you can't understand a Stirling engine simply by building one or watching one operating from the outside: you need to think hard about the cycle of steps it's going through, what's going on with the gas inside, and how that differs from what happens in an ordinary steam engine.
Anyway, let's see if we can explain the Stirling engine properly, first by looking at the parts it contains, then thinking about what they do, and finally looking at a bit of the more complex (thermodynamic) theory.

Photo: A prototype Stirling engine developed by NASA c.1979. Photo courtesy of NASA Glenn Research Center and Internet Archive.
What are the key parts of a Stirling engine?
There are quite a few different designs of Stirling engines, and we'll consider one particular type known as a displacer (or displacement) Stirling engine (also known as a beta Stirling engine). These are the key parts:
Heat source
The heat source is where the engine gets all its energy from and it could be anything from a coal fire to a solar mirror concentrating the heat of the Sun (as in our top photo). Although described as external combustion engines, Stirling engines don't have to use combustion (actual burning of fuel) at all: they simply need a difference in temperature between the heat source (where the energy comes from) and the heat sink (where it ends up).
You can drive a small Stirling engine with the heat from a cup of coffee, the warm palm of someone's hand, or even (to many people's complete astonishment) with an ice cube: the energy the engine puts out comes from any difference in temperature between the heat source and the heat sink. Having said that, it's worth remembering that there's only so much work you can do with a tiny Stirling engine powered by something like a cup of coffee, simply because it contains only a relatively small amount of energy, which is very quickly used up.
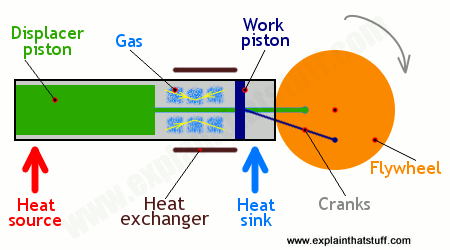
Artwork: The main parts of a displacer Stirling engine.
Gas
There's a volume of gas permanently sealed inside the machine in a closed cylinder. It can be ordinary air, hydrogen, helium, or some other readily available substance that remains a gas as it's heated and cooled through the engine's complete cycle (the repeated series of operations it goes through). Its only purpose is to move heat energy from the heat source to the heat sink, powering the piston that drives the machine, and then to go back again to pick up some more. The gas that moves heat is sometimes called the working fluid.
Heat sink
The place where the hot gas is cooled before being returned to the heat source. This is typically some sort of radiator (a piece of metal with fins attached), which expels waste heat to the atmosphere.
Pistons
There are various different types of Stirling engines, but I believe all of them have two pistons—that's one of the more obvious things that differentiates them from other engines. In a common design called a two-piston (or alpha) Stirling engine, there are two identical pistons and cylinders and the gas shuttles back and forth between them, heating and expanding, then cooling and compressing, before the cycle repeats.
In a different design, shown here, called a displacement (or beta) Stirling engine, there's one completely internal, piston called a displacer (colored green) whose job is to move the gas between the heat source and the heat sink. Unlike a normal piston in a steam engine, the displacer fits very loosely (with a little room to spare between the edge of the piston and the wall of the cylinder) and the gas flows around the outside of it as it moves back and forth. There's also a working piston (colored dark blue), which fits tightly into the cylinder, and turns the expansion of the gas into useful work that drives whatever the engine is powering. In bigger Stirling engines, the work piston usually has a heavy flywheel attached to build up momentum and keep the machine operating smoothly. The work piston and the displacer piston are both constantly moving, but they are out of step (one quarter cycle or 90° out of phase) with one another; they're powered by the same wheel but the displacer piston is always one quarter cycle (90°) ahead of the work piston.
Heat exchanger
Also known as the regenerator, the heat exchanger sits in the closed chamber between the heat source and the heat sink. As the hot gas moves past the regenerator, it gives up some of its heat, which the regenerator holds onto. When the gas moves back, it picks up this heat again. Without the regenerator, this heat would be lost to the atmosphere and wasted. The heat exchanger greatly improves the efficiency and power of the engine. Some Stirling engines have multiple heat exchangers.